In this article, we will learn the periodic table mnemonics. We can learn the periodic table with mnemonic thus making it easy to memorise the whole periodic table at once. Before starting, let’s take a quick glance of mnemonic and its use in chemistry.
Mnemonics are one of the easiest ways to learn something. We can use mnemonics in chemistry to remember the periodic table, reactivity series, etc.
The modern periodic table has 7 horizontal rows called periods and 18 vertical columns called groups. It was given by Henry Moseley.
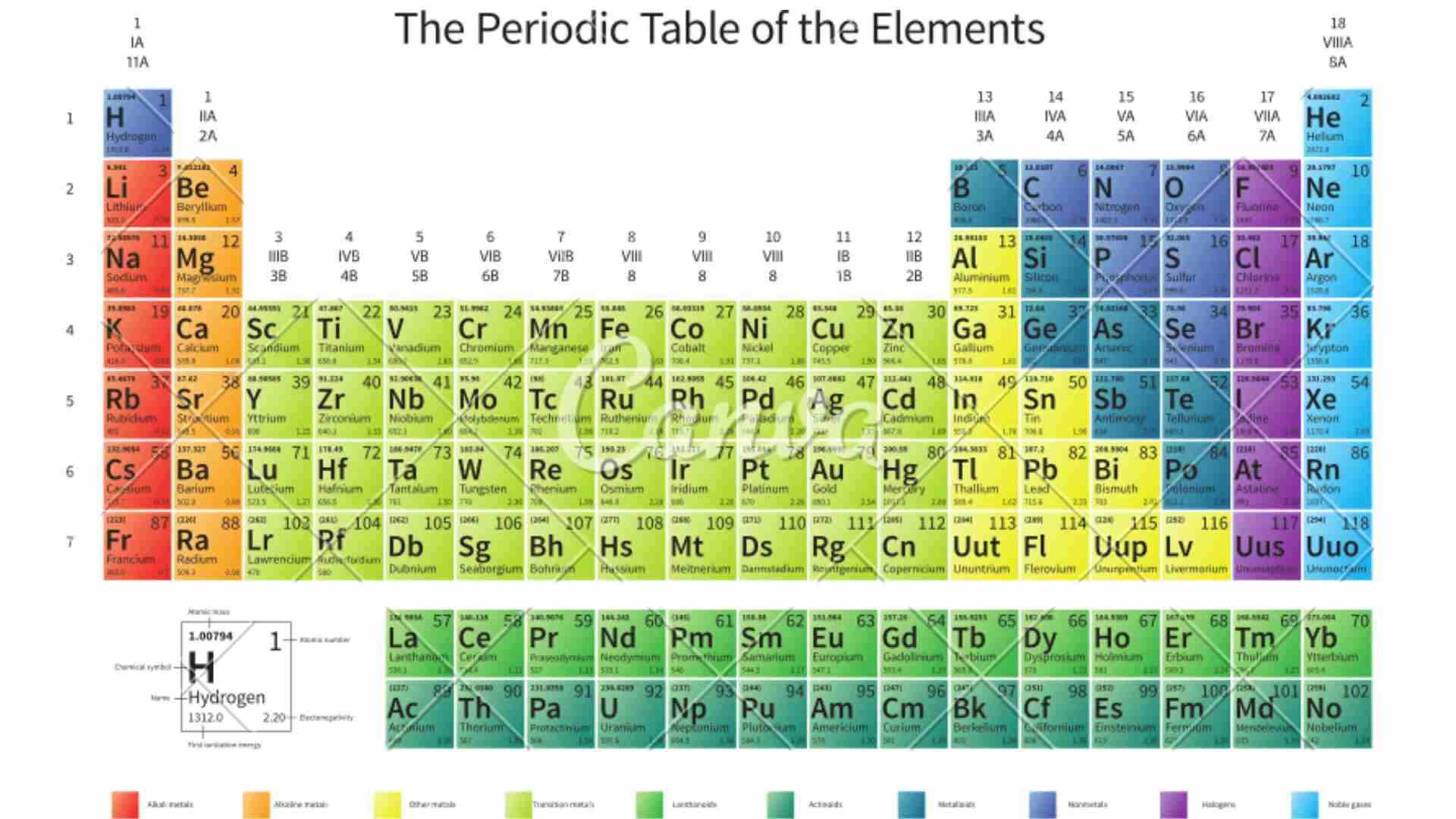
Periodic Table Mnemonics for s-block (Group 1 and 2) and p-block (Group 13-18) elements
So guys, let’s start:
Remembering the order of the first 10 elements of the periodic table is a child’s play! Just memorize the sentence given below and you would be able to memorize it easily. The chemical symbol of each of these elements is given in bold type.
The first 10 elements are as follows- Hydrogen (H), Helium (He), Lithium (Li), Beryllium (Be), Boron (B), Carbon (C), Nitrogen (N), Oxygen (O), Fluorine (F), Neon (Ne).
Mnemonic: Happy Henry Likes Beans Brownies and Chocolates Nuts Over Friday News.
Group 1 and Group 2 elements are collectively called as s-block elements. They have similar physical and chemical properties. Group 1 elements have 1 valence electron in their outermost shell and Group 2 elements have 2 valence electrons in their outermost shell.
Group 1 (also known as Alkali Metal): Lithium (Li), Sodium (Na), Potassium (K), Rubidium (Ru), Caesium (Cs), and Francium (Fr).
Mnemonic: LiNa Ki Ruby Cse Friyaad
Group 2 (also known as Alkaline Earth Metal): Beryllium (Be), Magnesium (Mg), Calcium (Ca), Strontium (Sr), Barium (Br), and Radium (Ra).
Mnemonic: Behen Maange Car, Scooter Brother Razi
The group 13 to group 18 elements are collectively called as p-block elements. It has 6 groups included in it.
The group 13 elements are called the Boron group or the Icosagens or Triels. They have 3 valence electrons in their outermost shell.
Group 13: Boron (B), Aluminium (Al), Gallium (Ga), Indium (In), and Thallium (Tl)
Mnemonic: Besides All, the Games is In sTill
The group 14 elements are called the Carbon group or Crystallogens, Tetragens or Tetrels. They have 4 valence electrons in their outermost shell.
Group 14: Carbon (C), Silicon (Si), Germanium (Ge), Tin (Sn), and Lead (Pb).
Mnemonic: Chemistry Sir Gives Standard Problems.
The group 15 elements are called the Nitrogen group or Pnictogens. They have 5 valence electrons in their outermost shell and has valency 3.
Group 15: Nitrogen (N), Phosphorus (P), Arsenic (As), Antimony (Sb), and Bismuth (Bi).
Mnemonic: Nahi Pasand Aise Sab Bindhi.
The group 16 elements are called the Oxygen group or Chalcogens. They have 6 valence electrons in their outermost shell and has valency 2.
Group 16: Oxygen (O), Sulphur (S), Selenium (Se), Tellurium (Te), and the radioactive element Polonium (Po).
Mnemonic: Om Sharma Se TePo
The group 17 elements are called Halogens. They have 7 valence electrons in their outermost shell and has a valency 1.
Group 17: Fluorine (F), Chlorine (Cl), Bromine (Br), Iodine (I), and Astatine (At).
Mnemonic: First CBI Attacked
The group 18 elements are called Noble gases, excluding Helium. They have 8 valence electrons in their outermost shell and has valency 0 and thus having an octet.
Group 18: Helium (He), Neon (Ne), Argon (Ar), Krypton (Kr), Xenon (Xe), and the radioactive Radon (Rn).
Mnemonic: He Never Arrived, Karo Xerox Rozana